|
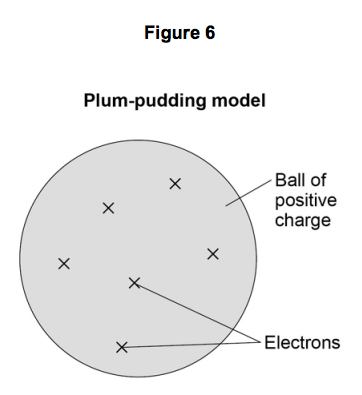
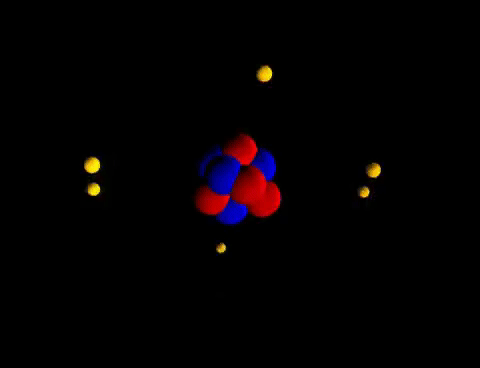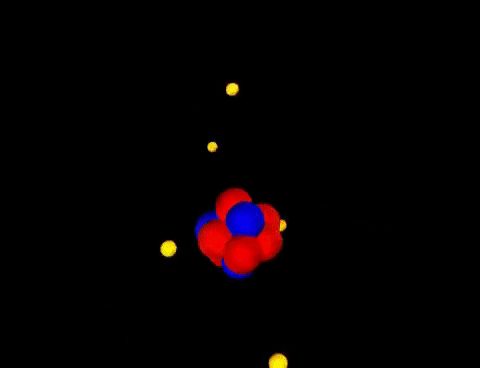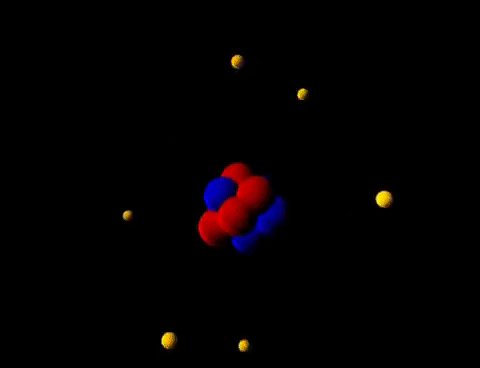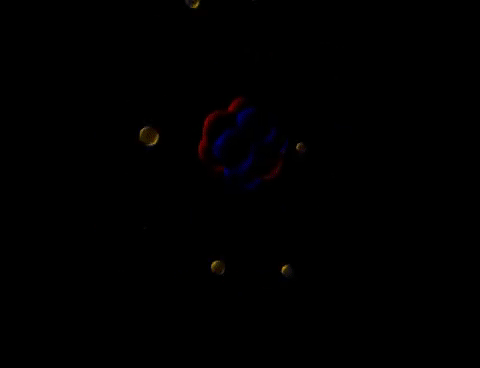
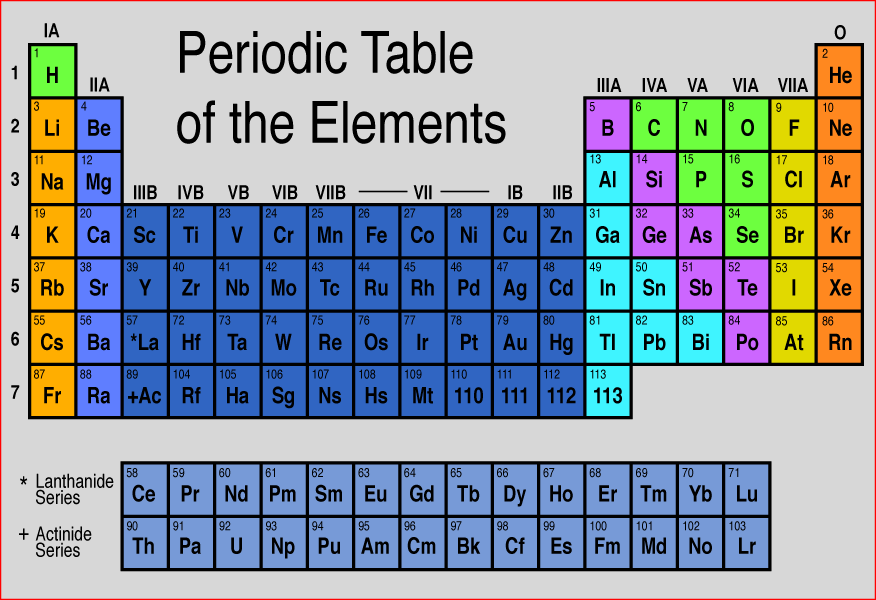
Matter is everything that we can see, smell, touch, feel and even can't see. Everything is matter, there isn't anything that isn't matter. The smallest unit of matter is called an atom. Who figured this out? And why??! If I chop a piece of metal into a bazillion-gillian pieces and the smallest, tiniest piece I'm left with that is still that piece of metal...this could be compared to an atom. John Dalton was the first scientist to suggest that elements are made of tiny particles that can't be destroyed. He believed that particles could be so small you couldn't see them. He even called them atoms. Along came Sir Joseph John (J. J.) Thomson who thought that there must be negatively charged particles in atoms AND they were nestled beside positively charged particles inside the atom. Imagine raisins in an oatmeal-raisin cookie.... He called it the "Plum-pudding" Model. Yick! Oatmeal-raisin cookies are much tastier. Then another inspired scientist, Ernest Rutherford envisioned each atom had a heavy positively charged center, which he named "nucleus". He decided the negatively charged particles were circling around the outside of the atom. Mr. Rutherford had student (that was trying to get the best grades possible), Sir James Chadwick that proposed that the uncharged particles in the nucleus were called "neutrons". His favorite student: James Chadwick Together, yet not together, these scientists figured out a problem on their own. Their work tied together like a web, that led others to create the modern Atomic Model that we know today. These heavy thinkers and seers of empty space figured out that: 1. Matter is anything that has mass and takes up space. 2. Atoms are the smallest units of matter. 3. Atoms cannot be divided...(only smashed in particle accelerators...) Remember how we have had to make models of the Seasons, and Eclipses, and the Solar System, and Planets...Yep...we are going to study models of matter: Atomic Models. Technology has been able to capture some of those tiniest pieces of matter with electron microscopes. Class! Meet some atoms! You can find all of the atoms we know about on this handy table: We don't memorize the elements in the 6th grade...but wait, it's coming!
13 Comments
kajetan
2/5/2019 04:20:10 pm
i can cut wood better than that one guy
Reply
Quinton Jones
2/5/2019 06:06:23 pm
Sure
Reply
kajetan
2/5/2019 04:23:02 pm
first to comment at 4:20
Reply
Quinton Jones
2/5/2019 06:05:48 pm
Imagine the people next to that one girl who all they were hearing is chew chew chew chew chew
Reply
Brooklyn.T
2/5/2019 07:25:09 pm
I wonder how much matter is in the world
Reply
Zach
2/5/2019 08:15:03 pm
Atoms are really cool.
Reply
danny
2/5/2019 08:25:25 pm
i bet thats waht charlie sounds like when she chews
Reply
ian
2/5/2019 08:51:10 pm
every time i eat gum i feel like
Reply
ian
2/5/2019 08:52:26 pm
im chewing too loud
Reply
ian
2/5/2019 08:53:27 pm
hehehehehehehehehehehehehehehe
Reply
OLIVER ROPER
2/5/2019 09:36:46 pm
That one guy looks like the "THAT'S A LOT OF DEMAGE" dude and he is pretty LIT. he heheheh heheheh hehe
Reply
Andrew
2/5/2019 10:07:46 pm
Nothing sponge bob I am just looking at science
Reply
Leave a Reply. |
Mrs. TaylorI love science! Everything about the world is interesting and never boring. I love to study plants, animals, insects, and people. My favorite subjects are my students who are the most unique organisms on the planet! Categories |







 RSS Feed
RSS Feed
