|
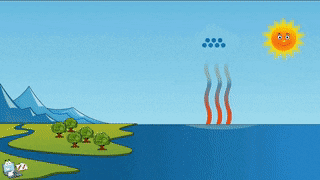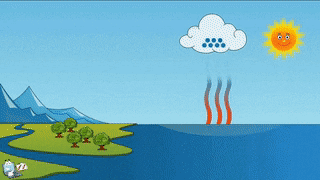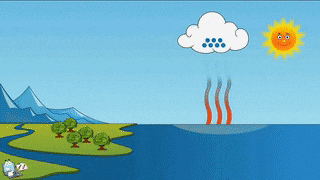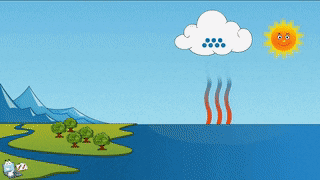
As a teacher I am obligated to give you the evidence that evaporation and sublimation are not the same. Yes, the verbage can be confusing, but evaporation and sublimation are different. Sublimation and evaporation are similar in that they are both phase changes where a substance changes to a gas...but that's where the similarity ends. Sublimation comes from a solid changing to a gas, and evaporation is liquid changing to a gas. Hopefully you can tell the difference between the two examples...if not, oh man, we got to go way back to the beginning and start over.... What we need for evaporation is a heat source, like the sun, or a room with a big window that the sun can shine through, or a big room with a big heater blasting warm air into it....and water, like a lake, ocean or puddle. For sublimation we need a solid gas that has been super frozen and kept super frozen. Dry ice doesn't need a heat source to trigger it to sublimate. A warm room, a refrigerator, a freezer is warm enough to sublimate dry ice. To turn dry ice into a liquid, we need pressure. Where would this pressure come from? According to the kinetic theory (particles moving), we need something that would create so much pressure, the gas atoms would be squeezed so close together that they would begin to flow. This kind of pressure could be found in a gas planet that has a liquid core. The gases are compressed by the mass of the planet and gravity, to make liquid. Do scientists know this for sure? No, but theoretically, this is possible. Dry ice sublimates at -109 degrees Fahrenheit. Our classroom is about 72 degrees Fahrenheit. For dry ice, that's a piece of cake to sublimate and it will, quickly!
Is this clear? Claaaaasssssssss??
13 Comments
|
Mrs. TaylorI love science! Everything about the world is interesting and never boring. I love to study plants, animals, insects, and people. My favorite subjects are my students who are the most unique organisms on the planet! Categories |






 RSS Feed
RSS Feed
