|
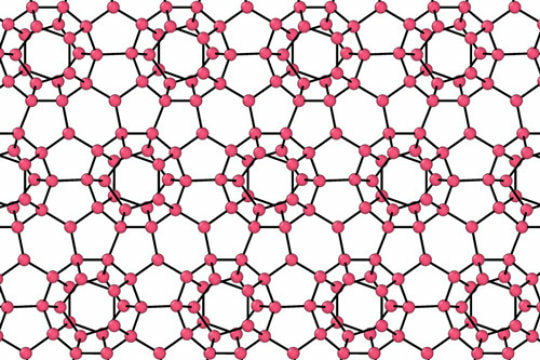
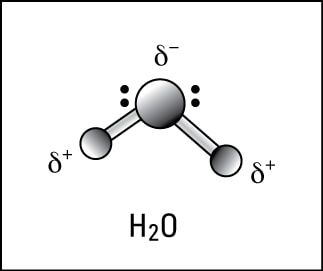
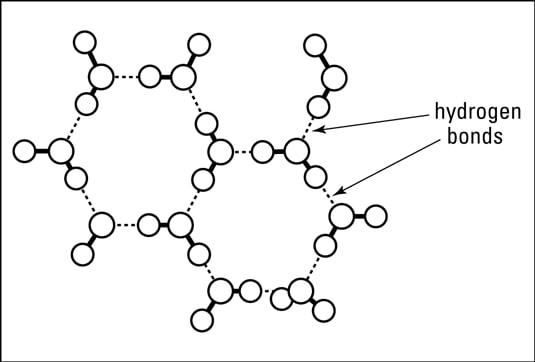
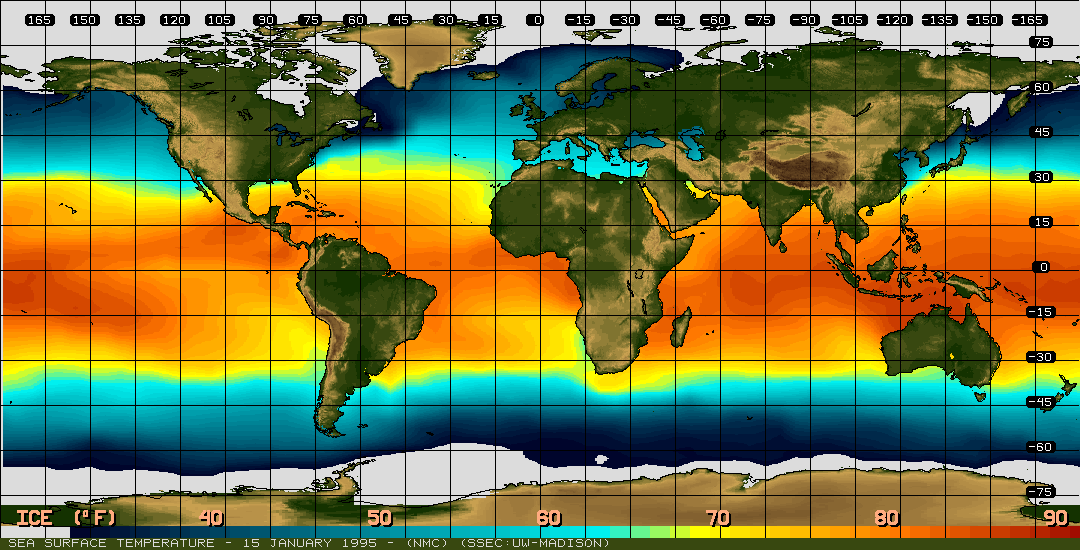
Water has fascinated scientists, children and others for years. We swim in it, drink it, ski on it, sip it, water our plants with it...I mean, we do stuff with water endlessly, every single day! Yet, water is a mystery. Water has unique behaviors that are still not understood. Water becomes thicker when compressed: Bugs and birds can walk on top of water... What gives? Why does water act so differently compared to other liquids? Molecular structure. Simple. Water molecules are like teeny magnets, they have a positive end and a negative end. So they are HUGELY attractive to other molecules or atoms and HUGELY repellent with other atoms and molecules. Because water is so POLAR the bonds it makes with other atoms and molecules is very strong (speaking on a molecular level...). Trying to break water bonds, or hydrogen bonds is difficult and that's one reason water is so mystifying.... When water freezes those hydrogen bonds LOCK into place making a tight crystal-like structure with air pockets in between the oxygen and the hydrogen. The holes make the "ice" expand and increases the size of the ice, yet the ice is less dense than it was as a liquid. Water absorbs LOTS of energy from the Sun and keeps water warm. If you look at thermal energy in the ocean you can see how the Sun heats the oceans near the equators and the warm waters travel in currents across the world providing us much needed weather! We need water to be strange and unique. If it wasn't, life might not be here...
15 Comments
|
Mrs. TaylorI love science! Everything about the world is interesting and never boring. I love to study plants, animals, insects, and people. My favorite subjects are my students who are the most unique organisms on the planet! Categories |







 RSS Feed
RSS Feed
